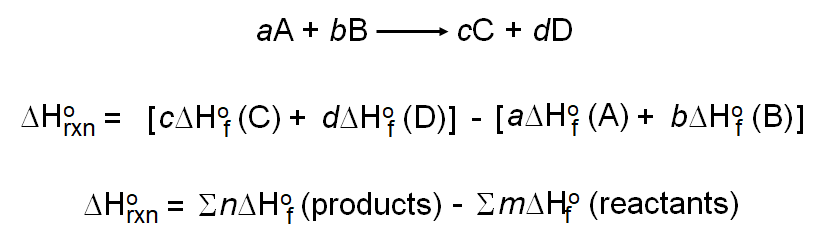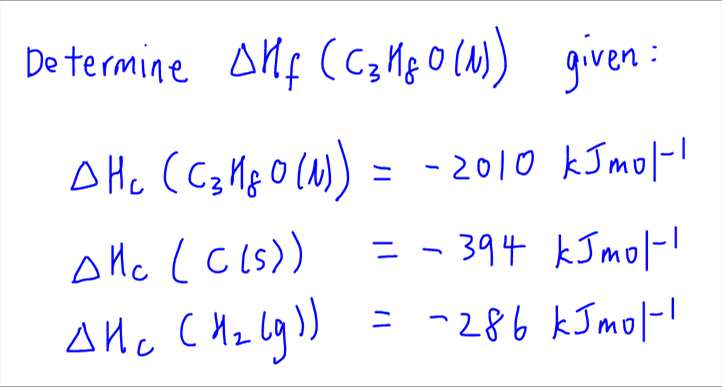By the end of today's lesson you should · know what enthalpy is · · · understand the difference between a molar enthalpy v
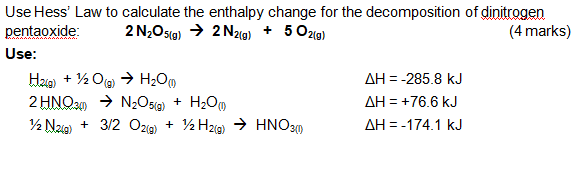
Use Hess' Law to calculate the enthalpy change for the decomposition of dinitrogen pentaoxide? | Socratic

Calculate the enthalpy change for the process CCl4(g)→ C(g) + 4Cl(g) and calculate bond enthalpy of C - Cl in CCl4(g) Δ vapH^ (CCl4) = 30.5 kJ mol ^-1 . Δ fH^ (

Calculate the enthalpy change for the process CCl4(g)→ C(g) + 4Cl(g) and calculate bond enthalpy of C - Cl in CCl4(g) Δ vapH^ (CCl4) = 30.5 kJ mol ^-1 . Δ fH^ (
16. Calculate the enthalpy change for the process CCl4(g)————C(g)+4Cl(g) And calculate bond enthalpy of C Cl in CCl4(g). Δ vapH(CCl4)=30.5 kj /mol Δ fH(CCl4)= 135.5 kj/mol Δ aH(C)=715.0 kj/mol Δ aH(Cl2)=242 kj/mol

Calculate the enthalpy change of 1 mole of reaction Na(s)+(1)/(2)Br(2)(g)rarrNaBr(s) in kcal Given : Delta H("sub")(Na)=137 kJ "mole"^(-1) , DeltaH("bond dissociation")(Br(2)(g))=144 kJ "mole"^(-1) Delta H("1 st ionisation")(Na(g)=496 kJ "mole"^(-1 ...



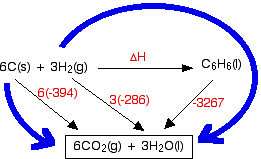


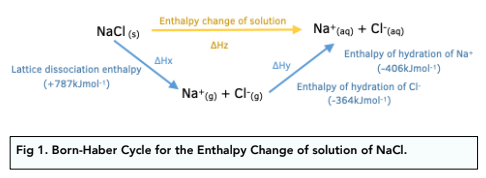
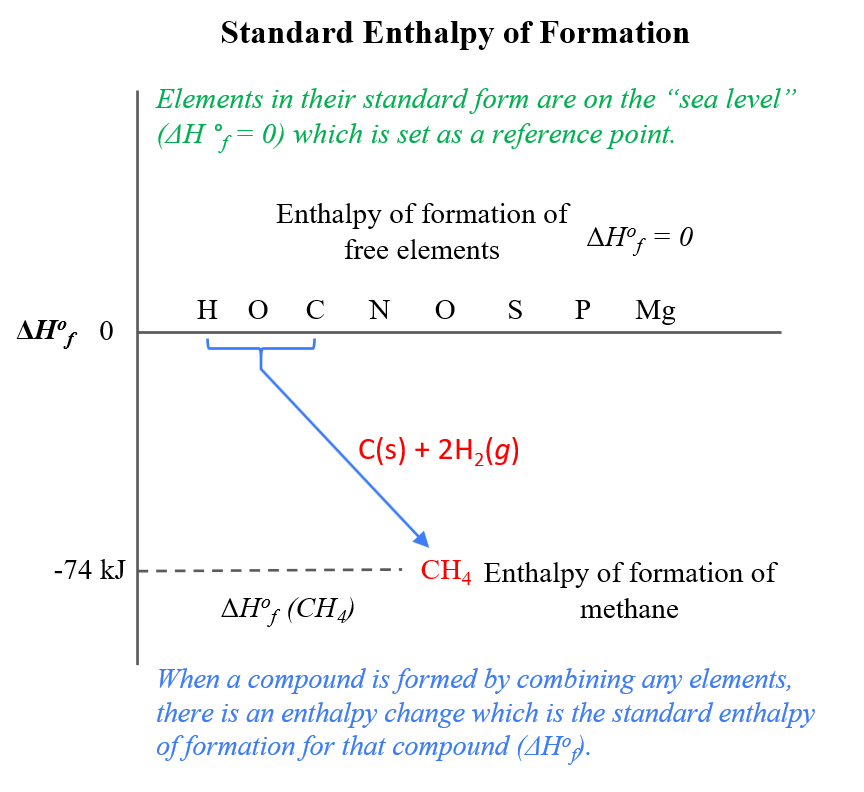

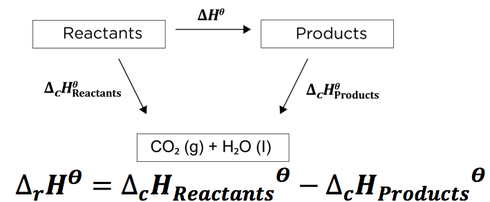
![Example] How to Calculate Enthalpy Change of a Reaction. - YouTube Example] How to Calculate Enthalpy Change of a Reaction. - YouTube](https://i.ytimg.com/vi/nmNQUGt6NiM/maxresdefault.jpg)