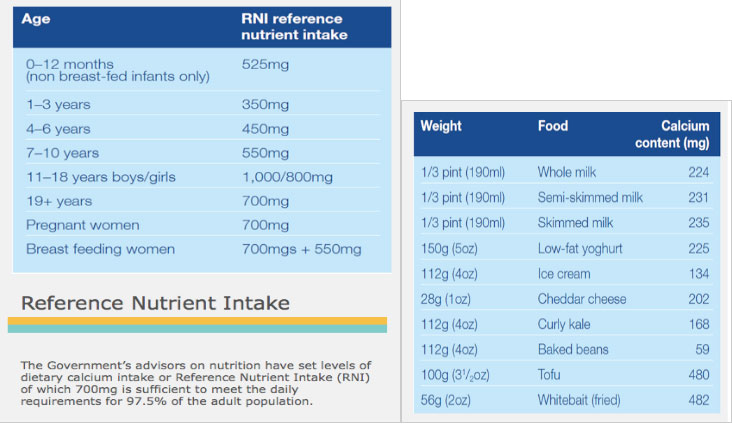
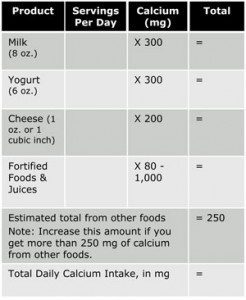
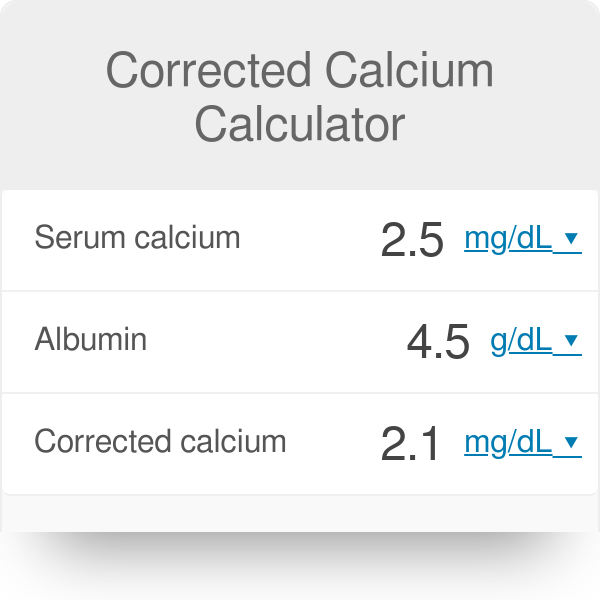
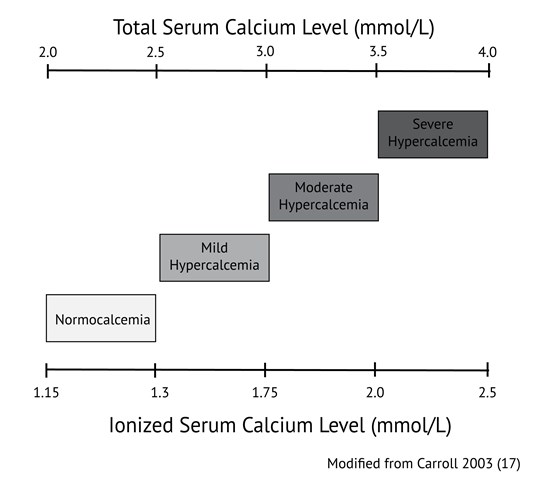
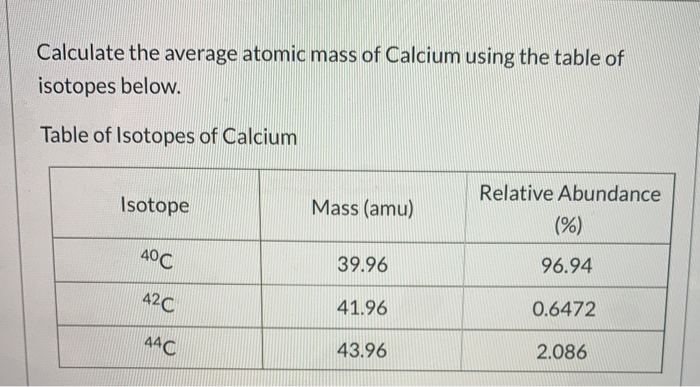
Things We Do for No Reason™: Calculating a “Corrected Calcium” Level - Kenny - 2021 - Journal of Hospital Medicine - Wiley Online Library

Calculate the mole ratio of `240 g` of calcium and `240 g` of magnesium. `(Ca = 40 , Mg = 24 u)` - YouTube

Calculate the mass per cent of each element present in the molecule of calcium carbonate - CBSE Class 9 Science - Learn CBSE Forum

Calculate the amount of calcium carbonate that would remain if 15 0 grams of calcium carbonate were reactedvwith 0 2 - Science - - 13298677 | Meritnation.com
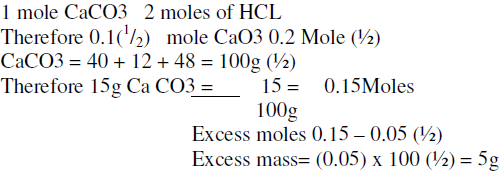
Calculate the amount of calcium carbonate that would remain if 15.0g of calcium carbonate were reacted with 0.2 moles of hydrochloric acid. The equation for the reaction...