
The relative atomic masses of two isotopes of an element are 15.9936 and 17.0036 amu. Calculate (A) the number of electrons (B) the number of protons (C) the number of neutrons (D)
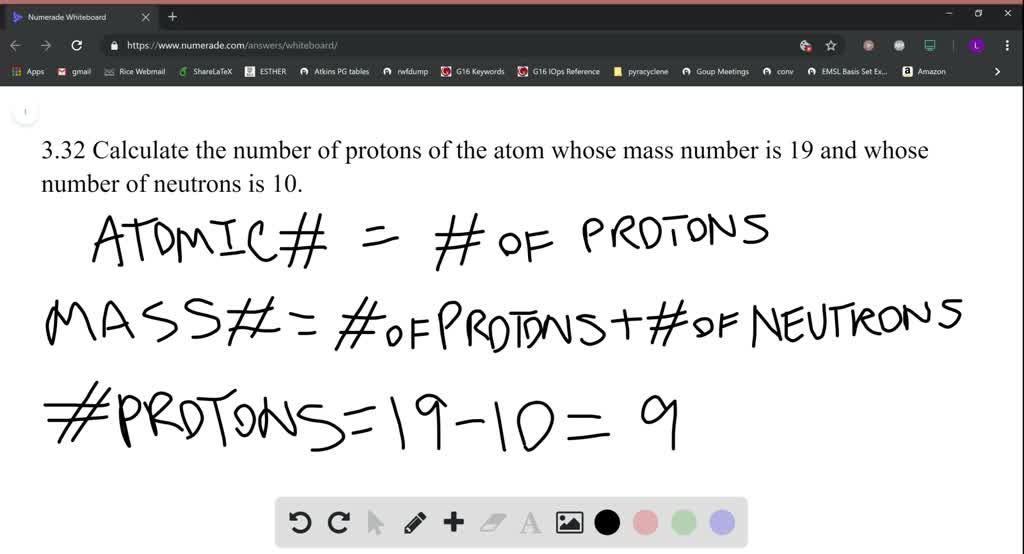
SOLVED:Calculate the number of protons of the atom whose mass number is 19 and whose number of neutrons is 10.




:max_bytes(150000):strip_icc()/atomic-mass--58dc0d885f9b58468332c41b.jpg)







.PNG)