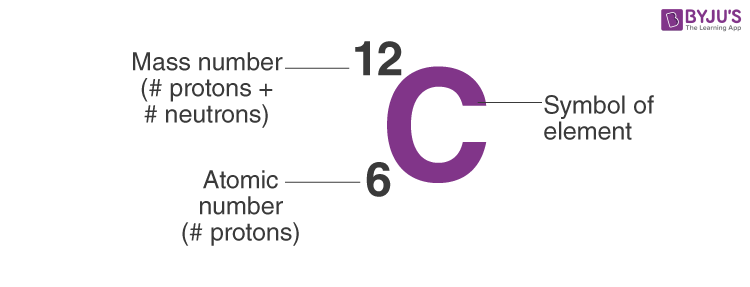
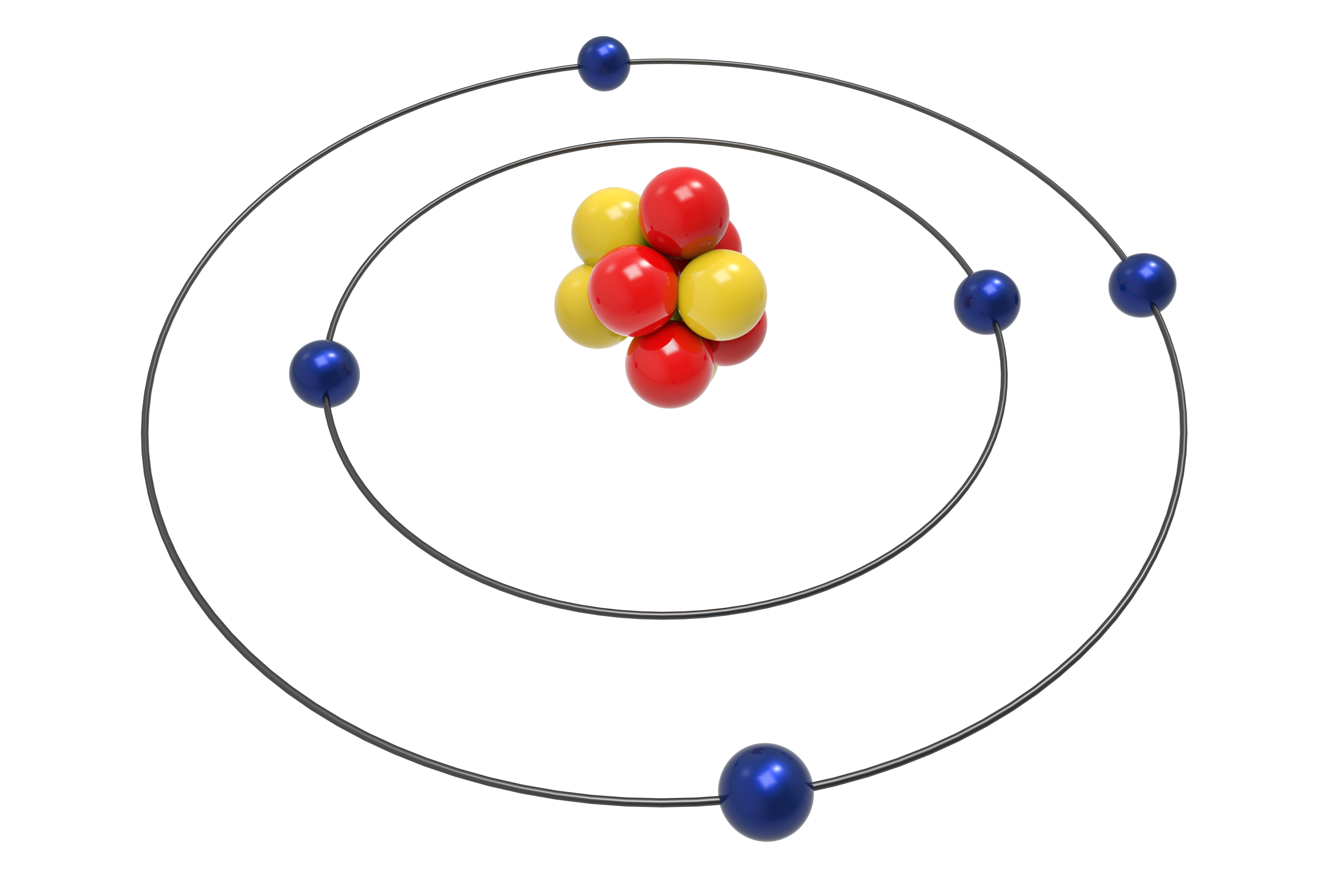
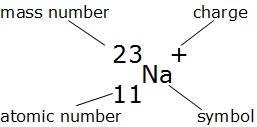
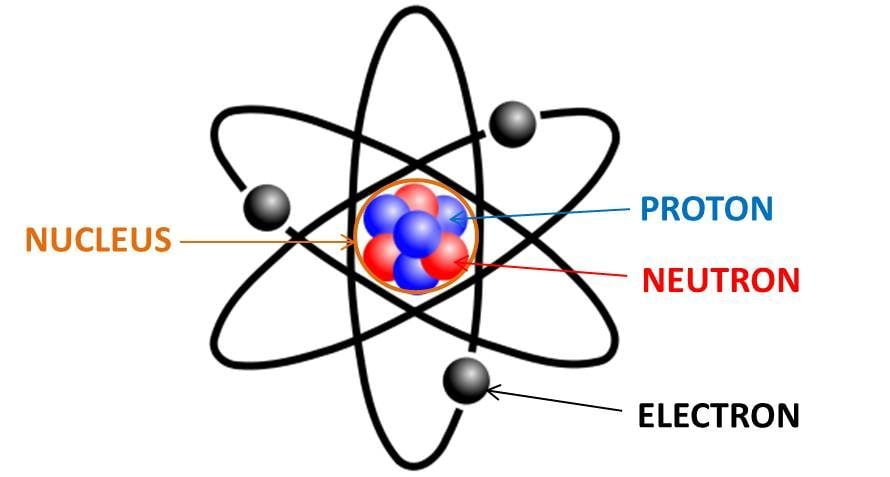
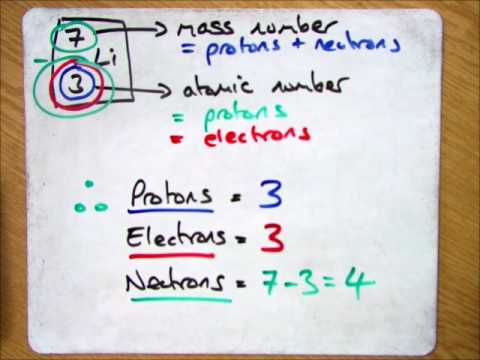
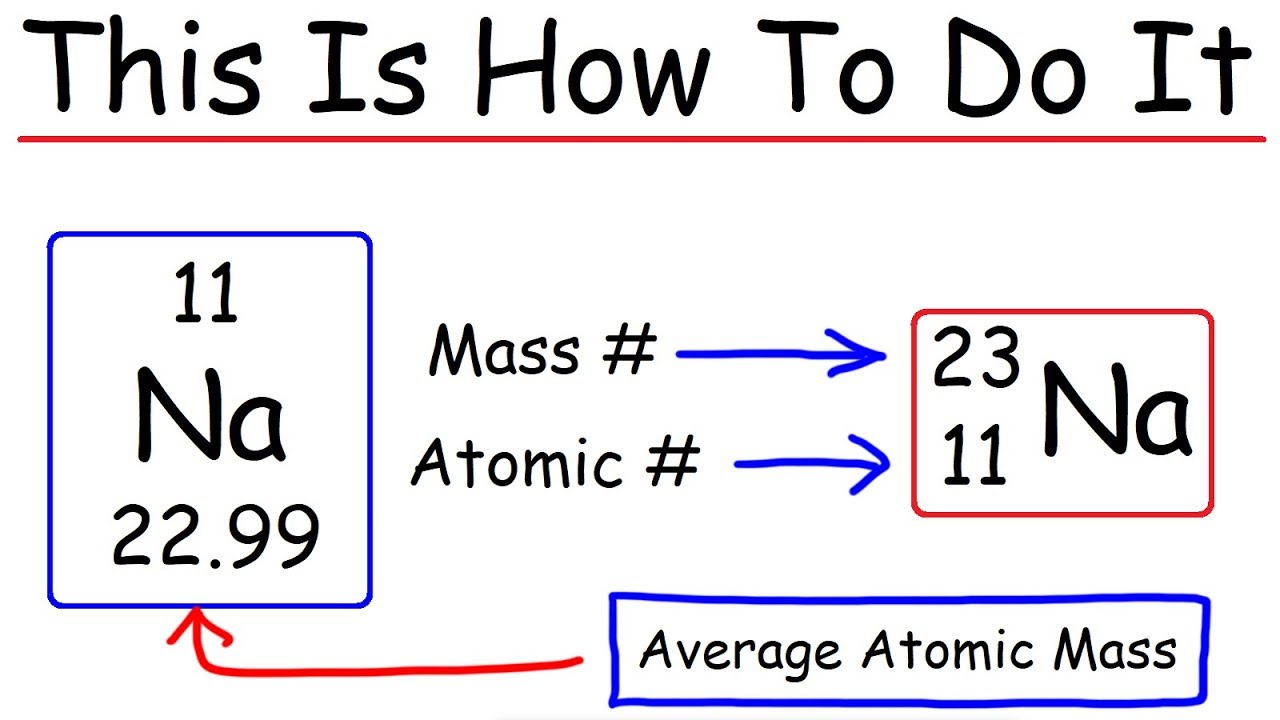
Calculate the number of electrons,protons,neutrons in 1.phosphorus atom. 2. Phosphate ion. MASS NO.=P=31, O=16 ATOMIC NO.P=15,O=8 along with explanation.
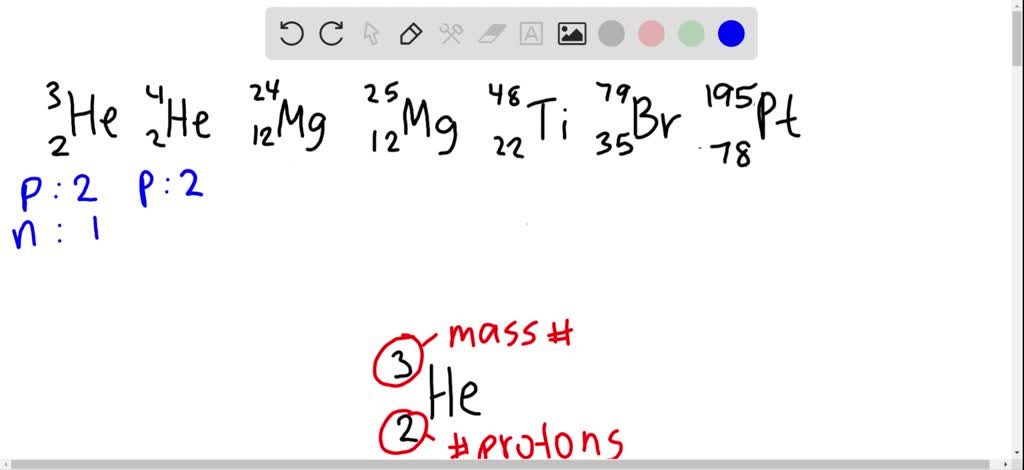
SOLVED:For each of the following species, determine the number of protons and the number of neutrons in the nucleus: 2^3 He,2^4 He,12^24 Mg,12^25 Mg,22^48 Ti,35^79 Br,78^195 Pt
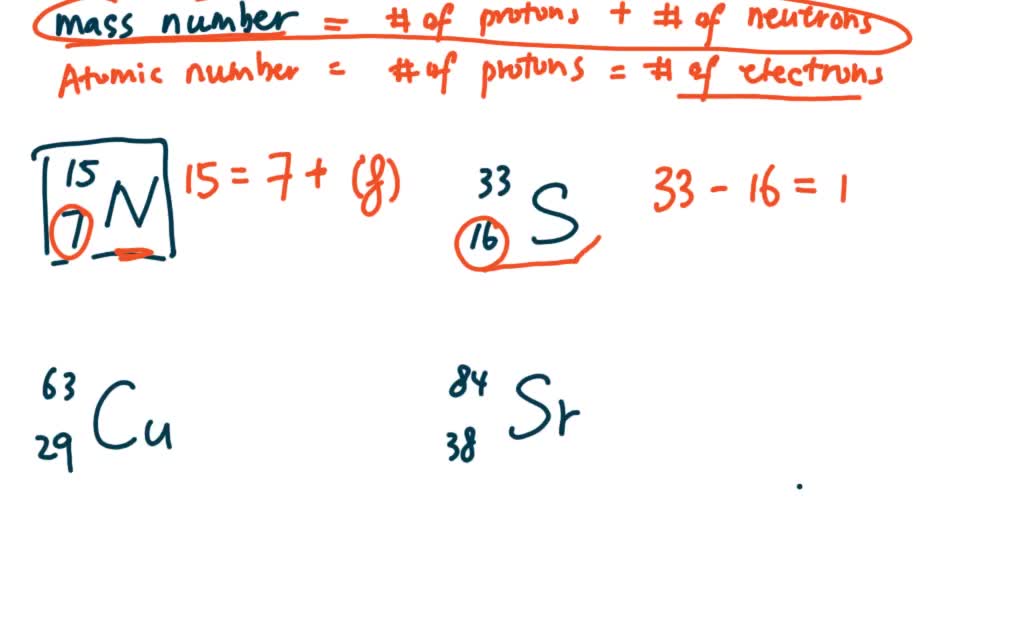
SOLVED:Indicate the number of protons, neutrons, and electrons in each of the following species: 7^15 N,16^33 S,29^63 Cu,38^84 Sr,56^130 Ba,74^186 W,80^202 Hg

i) Calculate the total number of electrons present in one mole of methane. (ii) Find (a) the total number and (b) the total mass of neutrons in 7 mg of ^14C .(Assume