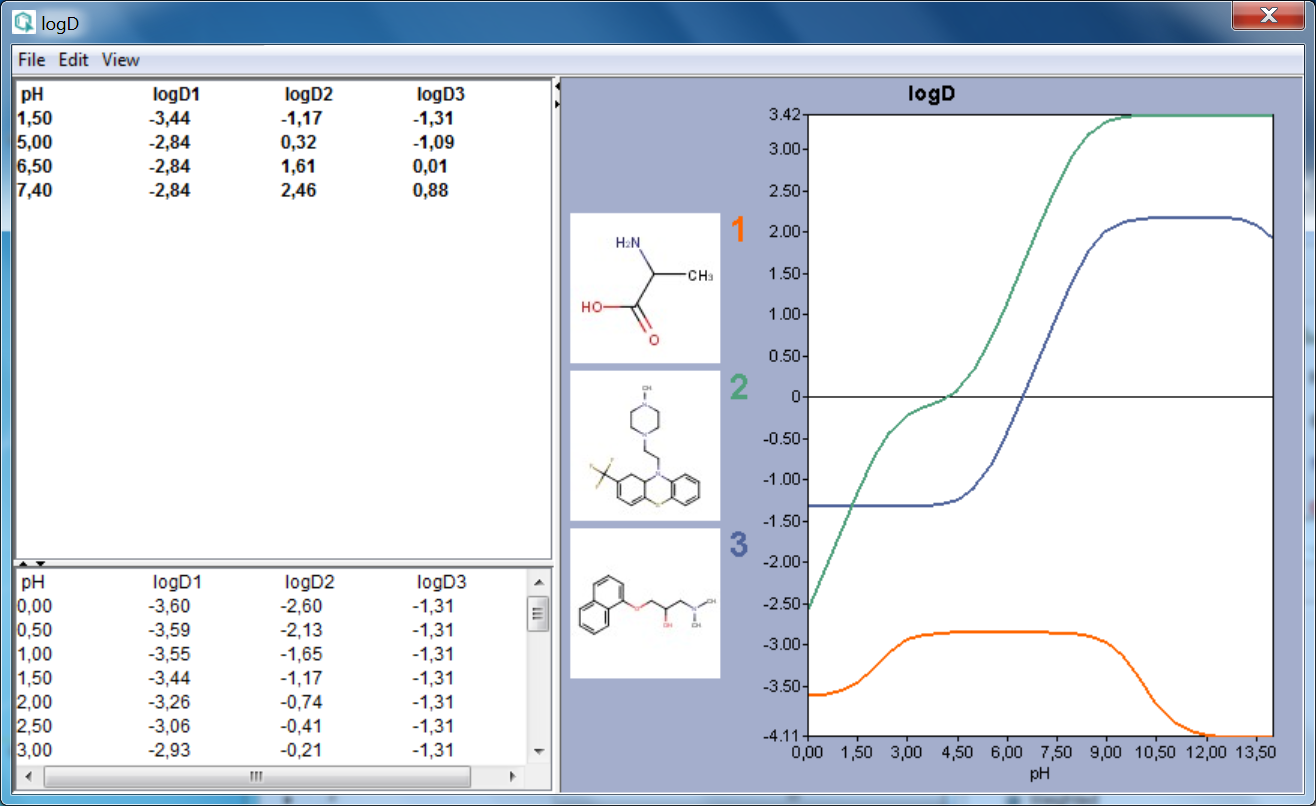
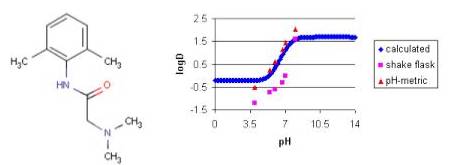
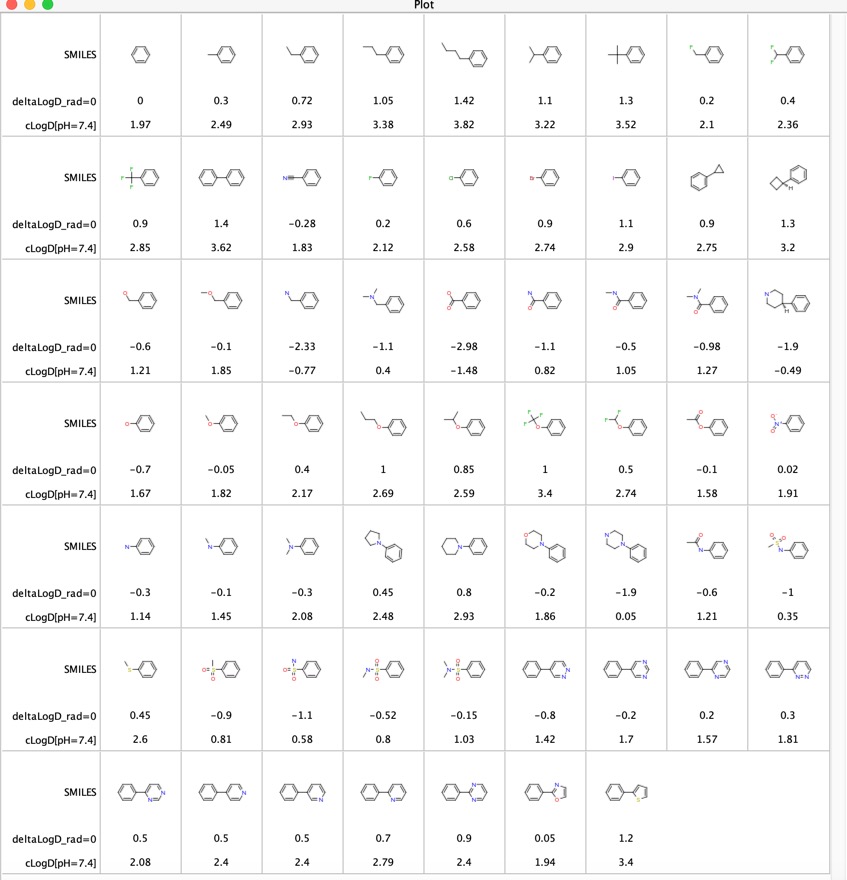
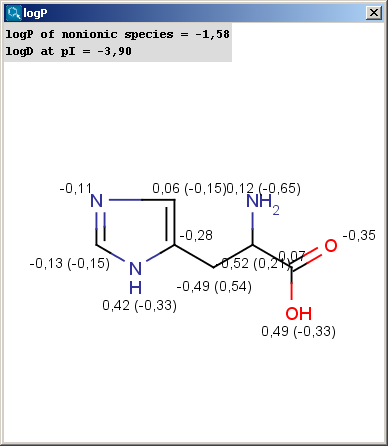
SOLVED: For an acidic drug, the following relationship between logD and logP exists when pH is much greater than pKa of the drug: LogD = LogP + (pH - pKa) Derive the

The Rule of Five Revisited: Applying Log D in Place of Log P in Drug-Likeness Filters | Molecular Pharmaceutics
Plot log D vs 1/T and the D(T) diffusion coefficient calculation of... | Download Scientific Diagram

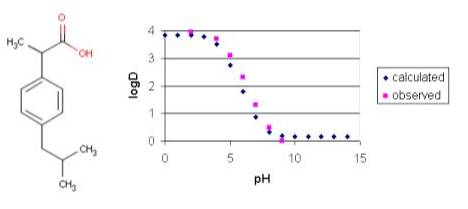



![chemaxoncalclogd [online documentation] chemaxoncalclogd [online documentation]](https://doc.mcule.com/lib/exe/fetch.php?w=800&tok=493d65&media=logd.png)