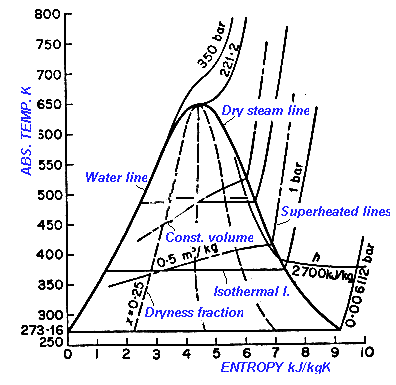
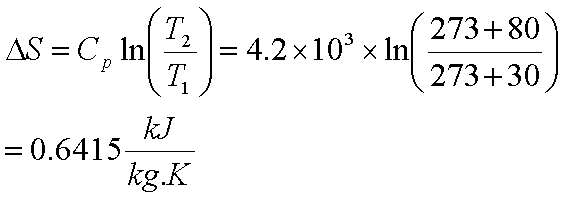Calculate the entropy change for vaporization of `1mol` of liquid water to stem at `100^()C`, if... - YouTube

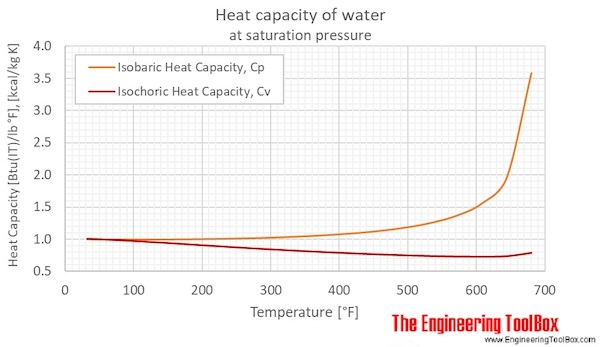
Calculate the enthalpy change of freezing of `1.0` mol of water at `10^()C` to ice at `-10^()C, ... - YouTube
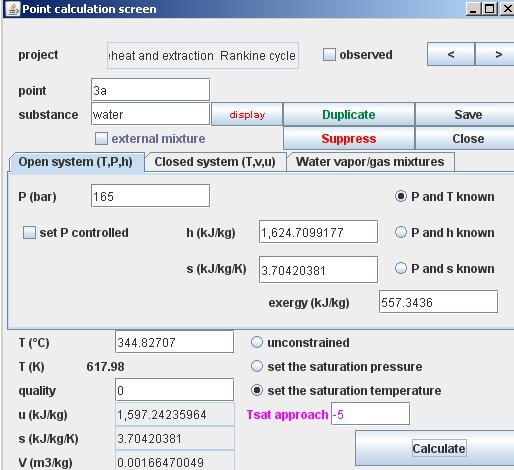
How to calculate entropy of water vapour in standard state (ideal gas) while vapourizing from liquid state at same temp. different pressure, and known standard entropy of water and enthalpy of vaporization -

Entropy Change For Melting Ice, Heating Water, Mixtures & Carnot Cycle of Heat Engines - Physics - YouTube
![Using some or all of the information below, calculate the standard molar entropy of I2 at 450 K. S^o = [{Blank}] J/K.mol at 450 K. | Homework.Study.com Using some or all of the information below, calculate the standard molar entropy of I2 at 450 K. S^o = [{Blank}] J/K.mol at 450 K. | Homework.Study.com](https://homework.study.com/cimages/multimages/16/screen_shot_2020-12-02_at_3.01.47_am7814899012014415578.png)